What Would Most Likely Happen to the Carbon Cycle if There Were No More Plants on Earth?
The Carbon Cycle
June 16, 2011
Carbon is the courage of life on Globe. We are made of carbon, nosotros consume carbon, and our civilizations—our economies, our homes, our ways of send—are built on carbon. We demand carbon, but that demand is likewise entwined with one of the most serious problems facing the states today: global climate change.


Carbon is both the foundation of all life on Earth, and the source of the majority of energy consumed by human culture. [Photographs ©2007 MorBCN (top) and ©2009 sarahluv (lower).]
Forged in the heart of aging stars, carbon is the fourth near abundant element in the Universe. Most of Earth'south carbon—about 65,500 billion metric tons—is stored in rocks. The rest is in the ocean, atmosphere, plants, soil, and fossil fuels.
Carbon flows betwixt each reservoir in an exchange called the carbon cycle, which has slow and fast components. Any change in the wheel that shifts carbon out of ane reservoir puts more carbon in the other reservoirs. Changes that put carbon gases into the temper result in warmer temperatures on World.
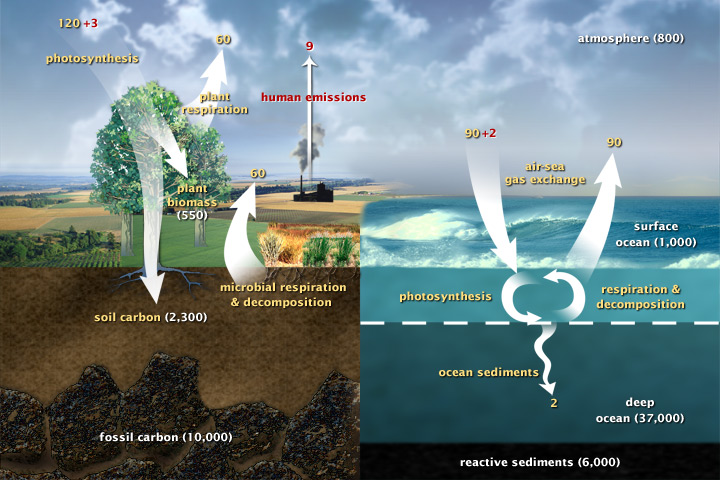
This diagram of the fast carbon cycle shows the movement of carbon between state, atmosphere, and oceans. Yellowish numbers are natural fluxes, and red are human contributions in gigatons of carbon per twelvemonth. White numbers bespeak stored carbon. (Diagram adapted from U.S. DOE, Biological and Ecology Enquiry Information Arrangement.)
Over the long term, the carbon cycle seems to maintain a residuum that prevents all of Globe'due south carbon from inbound the atmosphere (as is the example on Venus) or from being stored entirely in rocks. This balance helps keep Earth's temperature relatively stable, like a thermostat.
This thermostat works over a few hundred thousand years, every bit role of the slow carbon cycle. This means that for shorter fourth dimension periods—tens to a hundred thousand years—the temperature of Earth can vary. And, in fact, Earth swings between ice ages and warmer interglacial periods on these time scales. Parts of the carbon cycle may even dilate these short-term temperature changes.
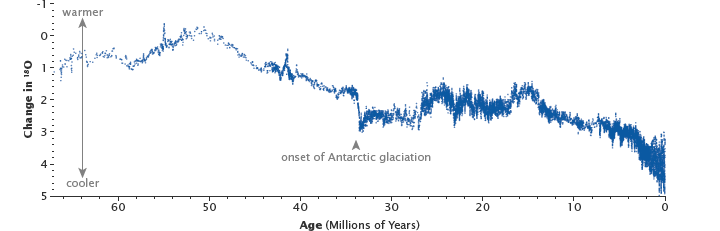
The uplift of the Himalaya, beginning 50 one thousand thousand years agone, reset Earth's thermostat by providing a big source of fresh rock to pull more carbon into the slow carbon cycle through chemical weathering. The resulting drop in temperatures and the formation of ice sheets changed the ratio between heavy and light oxygen in the deep bounding main, as shown in this graph. (Graph based on information from Zachos at al., 2001.)
On very long fourth dimension scales (millions to tens of millions of years), the movement of tectonic plates and changes in the rate at which carbon seeps from the Earth's interior may change the temperature on the thermostat. Earth has undergone such a change over the concluding fifty 1000000 years, from the extremely warm climates of the Cretaceous (roughly 145 to 65 1000000 years ago) to the glacial climates of the Pleistocene (roughly 1.8 meg to 11,500 years ago). [Encounter Divisions of Geologic Time—Major Chronostratigraphic and Geochronologic Units for more than information nearly geological eras.]
The Tiresome Carbon Cycle
Through a series of chemical reactions and tectonic activity, carbon takes betwixt 100-200 one thousand thousand years to motility between rocks, soil, bounding main, and atmosphere in the irksome carbon cycle. On boilerplate, 10xiii to 1014 grams (x–100 one thousand thousand metric tons) of carbon motility through the slow carbon cycle every year. In comparing, human emissions of carbon to the temper are on the order of 10xv grams, whereas the fast carbon bicycle moves ten16 to 1017 grams of carbon per year.
The movement of carbon from the atmosphere to the lithosphere (rocks) begins with rain. Atmospheric carbon combines with water to form a weak acid—carbonic acid—that falls to the surface in pelting. The acid dissolves rocks—a process called chemical weathering—and releases calcium, magnesium, potassium, or sodium ions. Rivers carry the ions to the ocean.

Rivers carry calcium ions—the effect of chemical weathering of rocks—into the ocean, where they react with carbonate dissolved in the water. The product of that reaction, calcium carbonate, is and so deposited onto the bounding main floor, where information technology becomes limestone. (Photo ©2009 Greg Carley.)
In the ocean, the calcium ions combine with bicarbonate ions to form calcium carbonate, the active ingredient in antacids and the chalky white substance that dries on your faucet if you live in an surface area with hard water. In the modern bounding main, most of the calcium carbonate is fabricated by beat-edifice (calcifying) organisms (such equally corals) and plankton (like coccolithophores and foraminifera). After the organisms die, they sink to the seafloor. Over time, layers of shells and sediment are cemented together and plow to stone, storing the carbon in stone—limestone and its derivatives.

Limestone, or its metamorphic cousin, marble, is rock made primarily of calcium carbonate. These rock types are often formed from the bodies of marine plants and animals, and their shells and skeletons can be preserved as fossils. Carbon locked upward in limestone can exist stored for millions—or even hundreds of millions—of years. (Photograph ©2008 Rookuzz (Hmm).)
Only 80 percent of carbon-containing rock is currently made this manner. The remaining 20 pct contain carbon from living things (organic carbon) that have been embedded in layers of mud. Estrus and pressure compress the mud and carbon over millions of years, forming sedimentary rock such equally shale. In special cases, when expressionless plant matter builds up faster than information technology can decay, layers of organic carbon go oil, coal, or natural gas instead of sedimentary rock like shale.

This coal seam in Scotland was originally a layer of sediment, rich in organic carbon. The sedimentary layer was eventually buried deep cloak-and-dagger, and the rut and pressure level transformed it into coal. Coal and other fossil fuels are a convenient source of energy, just when they are burned, the stored carbon is released into the atmosphere. This alters the remainder of the carbon bicycle, and is changing Earth'southward climate. (Photograph ©2010 Sandchem.)
The irksome cycle returns carbon to the atmosphere through volcanoes. Earth'due south state and ocean surfaces sit on several moving crustal plates. When the plates collide, ane sinks beneath the other, and the rock it carries melts under the extreme rut and pressure. The heated rock recombines into silicate minerals, releasing carbon dioxide.
When volcanoes erupt, they vent the gas to the atmosphere and cover the land with fresh silicate rock to brainstorm the bike once again. Now, volcanoes emit between 130 and 380 million metric tons of carbon dioxide per yr. For comparison, humans emit about 30 billion tons of carbon dioxide per twelvemonth—100–300 times more than than volcanoes—by called-for fossil fuels.
Chemistry regulates this trip the light fantastic toe between bounding main, state, and atmosphere. If carbon dioxide rises in the atmosphere because of an increment in volcanic action, for example, temperatures rise, leading to more rain, which dissolves more rock, creating more than ions that will eventually deposit more carbon on the ocean floor. It takes a few hundred yard years to rebalance the slow carbon bicycle through chemic weathering.

Carbon stored in rocks is naturally returned to the temper past volcanoes. In this photograph, Russian federation's Kizimen Volcano vents ash and volcanic gases in January 2011. Kizimen is located on the Kamchatka Peninsula, where the Pacific Plate is subducting beneath Asia. (Photograph ©2011 Artyom Bezotechestvo/Photo Kamchatka.)
However, the wearisome carbon cycle also contains a slightly faster component: the ocean. At the surface, where air meets water, carbon dioxide gas dissolves in and ventilates out of the ocean in a steady exchange with the atmosphere. Once in the body of water, carbon dioxide gas reacts with water molecules to release hydrogen, making the ocean more than acidic. The hydrogen reacts with carbonate from stone weathering to produce bicarbonate ions.
Before the industrial age, the sea vented carbon dioxide to the atmosphere in remainder with the carbon the ocean received during rock weathering. However, since carbon concentrations in the temper have increased, the sea at present takes more than carbon from the atmosphere than it releases. Over millennia, the sea will blot upwards to 85 pct of the extra carbon people have put into the atmosphere past burning fossil fuels, but the process is tedious because it is tied to the move of water from the body of water's surface to its depths.
In the concurrently, winds, currents, and temperature control the rate at which the bounding main takes carbon dioxide from the atmosphere. (Run across The Body of water's Carbon Balance on the Globe Observatory.) It is probable that changes in ocean temperatures and currents helped remove carbon from and and so restore carbon to the temper over the few thousand years in which the ice ages began and ended.
The Fast Carbon Cycle
The time it takes carbon to movement through the fast carbon cycle is measured in a lifespan. The fast carbon cycle is largely the movement of carbon through life forms on Earth, or the biosphere. Between 10xv and ten17 grams (1,000 to 100,000 meg metric tons) of carbon move through the fast carbon cycle every year.
Carbon plays an essential role in biology because of its power to form many bonds—upwardly to 4 per atom—in a seemingly endless diverseness of circuitous organic molecules. Many organic molecules contain carbon atoms that have formed strong bonds to other carbon atoms, combining into long chains and rings. Such carbon chains and rings are the basis of living cells. For case, DNA is made of two intertwined molecules built effectually a carbon concatenation.
The bonds in the long carbon chains contain a lot of energy. When the chains pause apart, the stored energy is released. This free energy makes carbon molecules an excellent source of fuel for all living things.
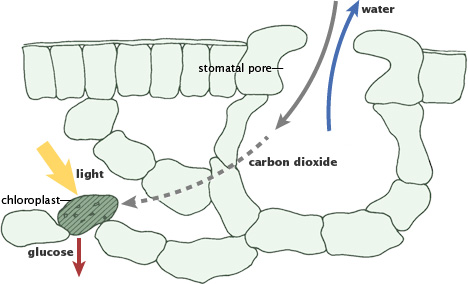
During photosynthesis, plants absorb carbon dioxide and sunlight to create fuel—glucose and other sugars—for building plant structures. This process forms the foundation of the fast (biological) carbon cycle. (Illustration adapted from P.J. Sellers et al., 1992.)
Plants and phytoplankton are the main components of the fast carbon wheel. Phytoplankton (microscopic organisms in the ocean) and plants take carbon dioxide from the atmosphere by arresting it into their cells. Using energy from the Dominicus, both plants and plankton combine carbon dioxide (CO2) and water to course sugar (CH2O) and oxygen. The chemical reaction looks like this:
CO2 + H2O + free energy = CHtwoO + O2
Iv things tin can happen to move carbon from a plant and render it to the temper, simply all involve the aforementioned chemical reaction. Plants suspension downward the sugar to become the energy they need to grow. Animals (including people) swallow the plants or plankton, and intermission down the institute saccharide to get energy. Plants and plankton die and decay (are eaten past leaner) at the cease of the growing season. Or burn down consumes plants. In each instance, oxygen combines with saccharide to release water, carbon dioxide, and energy. The basic chemic reaction looks similar this:
CH2O + O2 = CO2 + H2O + energy
In all four processes, the carbon dioxide released in the reaction unremarkably ends up in the atmosphere. The fast carbon cycle is so tightly tied to plant life that the growing flavour tin exist seen by the mode carbon dioxide fluctuates in the atmosphere. In the Northern Hemisphere winter, when few land plants are growing and many are decaying, atmospheric carbon dioxide concentrations climb. During the spring, when plants brainstorm growing once more, concentrations drop. It is equally if the Earth is breathing.
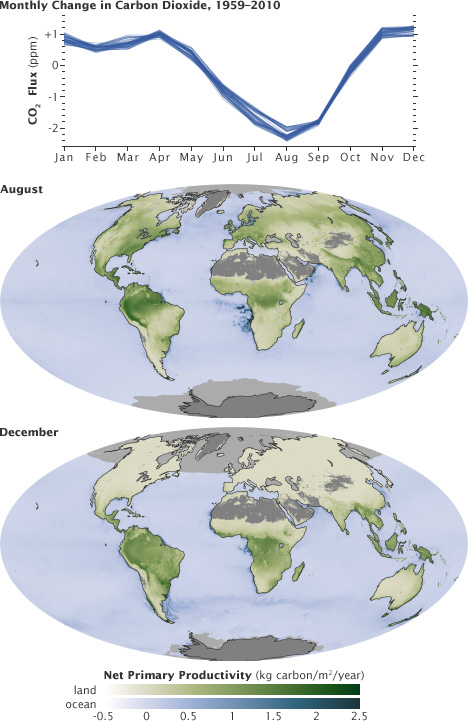
The ebb and flow of the fast carbon cycle is visible in the changing seasons. As the large state masses of Northern Hemisphere green in the bound and summertime, they describe carbon out of the temper. This graph shows the difference in carbon dioxide levels from the previous calendar month, with the long-term trend removed.
This wheel peaks in August, with nigh 2 parts per meg of carbon dioxide fatigued out of the atmosphere. In the fall and wintertime, as vegetation dies back in the northern hemisphere, decomposition and respiration returns carbon dioxide to the atmosphere.
These maps show net primary productivity (the amount of carbon consumed by plants) on country (light-green) and in the oceans (blue) during August and December, 2010. In August, the green areas of Due north America, Europe, and Asia correspond plants using carbon from the atmosphere to grow. In December, cyberspace primary productivity at high latitudes is negative, which outweighs the seasonal increase in vegetation in the southern hemisphere. As a event, the amount of carbon dioxide in the atmosphere increases.
(Graph by Marit Jentoft-Nilsen and Robert Simmon, using data from the NOAA Earth System Research Laboratory. Maps by Robert Simmon and Reto Stöckli, using MODIS data.)
Changes in the Carbon Cycle
Left unperturbed, the fast and dull carbon cycles maintain a relatively steady concentration of carbon in the atmosphere, land, plants, and ocean. Merely when anything changes the amount of carbon in one reservoir, the consequence ripples through the others.
In Globe's past, the carbon cycle has inverse in response to climate change. Variations in World'south orbit change the amount of free energy Earth receives from the Sun and leads to a cycle of ice ages and warm periods like Earth'south electric current climate. (See Milutin Milankovitch.) Ice ages developed when Northern Hemisphere summers cooled and ice built upwardly on country, which in turn slowed the carbon cycle. Meanwhile, a number of factors including cooler temperatures and increased phytoplankton growth may take increased the amount of carbon the ocean took out of the temper. The drop in atmospheric carbon caused additional cooling. Similarly, at the end of the concluding Ice Historic period, 10,000 years agone, carbon dioxide in the atmosphere rose dramatically as temperatures warmed.
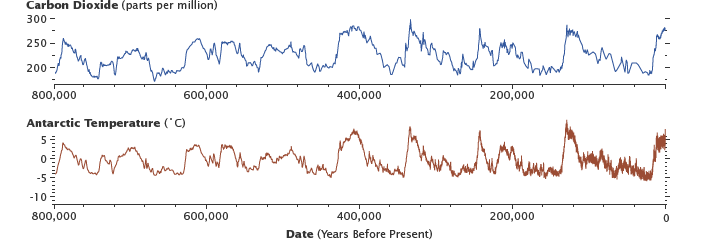
Levels of carbon dioxide in the atmosphere have corresponded closely with temperature over the past 800,000 years. Although the temperature changes were touched off past variations in Earth's orbit, the increased global temperatures released COii into the atmosphere, which in plough warmed the Earth. Antarctic ice-core information show the long-term correlation until about 1900. (Graphs past Robert Simmon, using information from Lüthi et al., 2008, and Jouzel et al., 2007.)
Shifts in Earth's orbit are happening constantly, in anticipated cycles. In nearly 30,000 years, Earth'southward orbit will have changed plenty to reduce sunlight in the Northern Hemisphere to the levels that led to the last ice age.
Today, changes in the carbon bike are happening because of people. We perturb the carbon cycle by burning fossil fuels and clearing land.
When we clear forests, we remove a dense growth of plants that had stored carbon in wood, stems, and leaves—biomass. Past removing a wood, we eliminate plants that would otherwise have carbon out of the atmosphere as they abound. We tend to replace the dense growth with crops or pasture, which store less carbon. We as well expose soil that vents carbon from rust-covered found affair into the atmosphere. Humans are currently emitting only under a billion tons of carbon into the atmosphere per year through state employ changes.

The called-for of fossil fuels is the main source of increased carbon dioxide in the temper today. (Photo ©2009 stevendepolo.)
Without human interference, the carbon in fossil fuels would leak slowly into the atmosphere through volcanic activity over millions of years in the slow carbon cycle. By called-for coal, oil, and natural gas, nosotros accelerate the process, releasing vast amounts of carbon (carbon that took millions of years to accumulate) into the atmosphere every year. Past doing so, we motility the carbon from the ho-hum bike to the fast cycle. In 2009, humans released about viii.4 billion tons of carbon into the atmosphere past called-for fossil fuel.
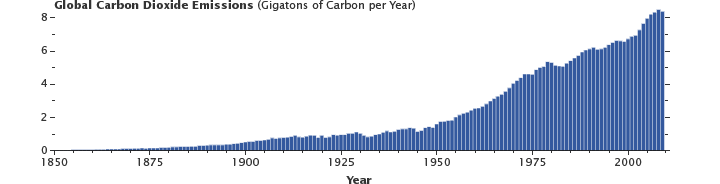
Emissions of carbon dioxide past humanity (primarily from the burning of fossil fuels, with a contribution from cement production) have been growing steadily since the onset of the industrial revolution. About one-half of these emissions are removed by the fast carbon cycle each yr, the rest remain in the temper. (Graph past Robert Simmon, using data from the Carbon Dioxide Information Analysis Center and Global Carbon Projection.)
Since the beginning of the Industrial Revolution, when people first started burning fossil fuels, carbon dioxide concentrations in the atmosphere accept risen from about 280 parts per one thousand thousand to 387 parts per million, a 39 percent increase. This means that for every million molecules in the atmosphere, 387 of them are now carbon dioxide—the highest concentration in two million years. Marsh gas concentrations accept risen from 715 parts per billion in 1750 to 1,774 parts per billion in 2005, the highest concentration in at least 650,000 years.
Effects of Changing the Carbon Cycle
All of this extra carbon needs to become somewhere. And so far, land plants and the ocean have taken upward about 55 percent of the actress carbon people have put into the atmosphere while about 45 percent has stayed in the atmosphere. Eventually, the land and oceans will take upward most of the extra carbon dioxide, only as much as 20 per centum may remain in the temper for many thousands of years.
The changes in the carbon cycle impact each reservoir. Excess carbon in the atmosphere warms the planet and helps plants on land grow more. Backlog carbon in the body of water makes the water more acidic, putting marine life in danger.
Temper
It is meaning that so much carbon dioxide stays in the atmosphere because CO2 is the well-nigh important gas for controlling Globe'due south temperature. Carbon dioxide, methane, and halocarbons are greenhouse gases that absorb a wide range of energy—including infrared energy (heat) emitted by the Earth—and then re-emit it. The re-emitted free energy travels out in all directions, but some returns to Earth, where information technology heats the surface. Without greenhouse gases, Earth would be a frozen -eighteen degrees Celsius (0 degrees Fahrenheit). With too many greenhouse gases, Earth would be like Venus, where the greenhouse atmosphere keeps temperatures around 400 degrees Celsius (750 Fahrenheit).

Rising concentrations of carbon dioxide are warming the atmosphere. The increased temperature results in higher evaporation rates and a wetter atmosphere, which leads to a vicious cycle of farther warming. (Photograph ©2011 Patrick Wilken.)
Because scientists know which wavelengths of energy each greenhouse gas absorbs, and the concentration of the gases in the atmosphere, they tin calculate how much each gas contributes to warming the planet. Carbon dioxide causes almost 20 percent of World's greenhouse effect; water vapor accounts for about 50 per centum; and clouds business relationship for 25 pct. The remainder is caused past small-scale particles (aerosols) and minor greenhouse gases like methane.
Water vapor concentrations in the air are controlled by Globe'due south temperature. Warmer temperatures evaporate more water from the oceans, aggrandize air masses, and lead to college humidity. Cooling causes water vapor to condense and fall out equally rain, sleet, or snowfall.
Carbon dioxide, on the other hand, remains a gas at a wider range of atmospheric temperatures than h2o. Carbon dioxide molecules provide the initial greenhouse heating needed to maintain water vapor concentrations. When carbon dioxide concentrations drop, Earth cools, some water vapor falls out of the atmosphere, and the greenhouse warming caused by water vapor drops. Also, when carbon dioxide concentrations ascent, air temperatures go upwards, and more h2o vapor evaporates into the temper—which then amplifies greenhouse heating.
And then while carbon dioxide contributes less to the overall greenhouse upshot than h2o vapor, scientists have constitute that carbon dioxide is the gas that sets the temperature. Carbon dioxide controls the amount of h2o vapor in the atmosphere and thus the size of the greenhouse event.
Rising carbon dioxide concentrations are already causing the planet to rut up. At the same time that greenhouse gases take been increasing, average global temperatures have risen 0.8 degrees Celsius (1.iv degrees Fahrenheit) since 1880.
This rise in temperature isn't all the warming we will see based on current carbon dioxide concentrations. Greenhouse warming doesn't happen right away because the ocean soaks up heat. This means that Earth'southward temperature will increment at least another 0.vi degrees Celsius (1 caste Fahrenheit) considering of carbon dioxide already in the atmosphere. The degree to which temperatures get upwardly across that depends in part on how much more carbon humans release into the atmosphere in the futurity.
Bounding main
About 30 percent of the carbon dioxide that people have put into the atmosphere has diffused into the ocean through the directly chemical exchange. Dissolving carbon dioxide in the ocean creates carbonic acid, which increases the acidity of the water. Or rather, a slightly alkaline metal ocean becomes a trivial less alkaline. Since 1750, the pH of the sea'due south surface has dropped by 0.1, a 30 pct change in acerbity.

Some of the excess COtwo emitted past man activeness dissolves in the sea, condign carbonic acid. Increases in carbon dioxide are not only leading to warmer oceans, just likewise to more acidic oceans. (Photo ©2010 Manner Out West News.)
Ocean acidification affects marine organisms in ii ways. Starting time, carbonic acrid reacts with carbonate ions in the water to form bicarbonate. However, those same carbonate ions are what shell-building animals similar coral need to create calcium carbonate shells. With less carbonate available, the animals need to expend more energy to build their shells. As a outcome, the shells stop up being thinner and more fragile.
Second, the more acidic h2o is, the better it dissolves calcium carbonate. In the long run, this reaction will allow the ocean to soak up backlog carbon dioxide considering more than acidic water volition deliquesce more than rock, release more than carbonate ions, and increase the ocean'southward capacity to absorb carbon dioxide. In the meantime, though, more than acidic h2o will dissolve the carbonate shells of marine organisms, making them pitted and weak.
Warmer oceans—a product of the greenhouse result—could also decrease the affluence of phytoplankton, which grow ameliorate in cool, food-rich waters. This could limit the ocean's ability to accept carbon from the atmosphere through the fast carbon wheel.
On the other manus, carbon dioxide is essential for plant and phytoplankton growth. An increase in carbon dioxide could increase growth by fertilizing those few species of phytoplankton and bounding main plants (like sea grasses) that accept carbon dioxide directly from the water. However, nigh species are not helped by the increased availability of carbon dioxide.
Country
Plants on state take taken up approximately 25 percent of the carbon dioxide that humans have put into the atmosphere. The amount of carbon that plants take up varies greatly from year to yr, but in general, the globe'south plants take increased the amount of carbon dioxide they absorb since 1960. Just some of this increase occurred as a direct result of fossil fuel emissions.
With more atmospheric carbon dioxide available to convert to plant matter in photosynthesis, plants were able to grow more than. This increased growth is referred to as carbon fertilization. Models predict that plants might grow anywhere from 12 to 76 percent more if atmospheric carbon dioxide is doubled, as long as naught else, like water shortages, limits their growth. Even so, scientists don't know how much carbon dioxide is increasing plant growth in the real world, because plants need more than carbon dioxide to abound.
Plants too demand water, sunlight, and nutrients, especially nitrogen. If a plant doesn't have one of these things, it won't grow regardless of how arable the other necessities are. In that location is a limit to how much carbon plants can take out of the atmosphere, and that limit varies from region to region. So far, information technology appears that carbon dioxide fertilization increases found growth until the plant reaches a limit in the amount of water or nitrogen available.
Some of the changes in carbon absorption are the result of land use decisions. Agriculture has become much more intensive, so we can grow more than food on less land. In high and mid-latitudes, abased farmland is reverting to forest, and these forests store much more carbon, both in forest and soil, than crops would. In many places, we preclude plant carbon from entering the temper past extinguishing wildfires. This allows woody material (which stores carbon) to build up. All of these land use decisions are helping plants blot homo-released carbon in the Northern Hemisphere.

Changes in land comprehend—forests converted to fields and fields converted to forests—have a corresponding effect on the carbon wheel. In some Northern Hemisphere countries, many farms were abandoned in the early on 20th century and the land reverted to forest. As a result, carbon was drawn out of the atmosphere and stored in copse on land. (Photograph ©2007 Husein Kadribegic.)
In the tropics, nevertheless, forests are being removed, oft through burn down, and this releases carbon dioxide. As of 2008, deforestation accounted for about 12 per centum of all human carbon dioxide emissions.
The biggest changes in the land carbon bike are likely to come because of climatic change. Carbon dioxide increases temperatures, extending the growing season and increasing humidity. Both factors have led to some additional plant growth. Yet, warmer temperatures besides stress plants. With a longer, warmer growing season, plants need more water to survive. Scientists are already seeing evidence that plants in the Northern Hemisphere deadening their growth in the summer because of warm temperatures and h2o shortages.
Dry, water-stressed plants are too more susceptible to burn and insects when growing seasons go longer. In the far northward, where an increase in temperature has the greatest touch on, the forests have already started to fire more than, releasing carbon from the plants and the soil into the temper. Tropical forests may also exist extremely susceptible to drying. With less water, tropical trees slow their growth and take upward less carbon, or die and release their stored carbon to the temper.
The warming caused past rising greenhouse gases may likewise "broil" the soil, accelerating the rate at which carbon seeps out in some places. This is of particular business organisation in the far north, where frozen soil—permafrost—is thawing. Permafrost contains rich deposits of carbon from institute matter that has accumulated for thousands of years considering the cold slows decay. When the soil warms, the organic matter decays and carbon—in the form of marsh gas and carbon dioxide—seeps into the atmosphere.
Current research estimates that permafrost in the Northern Hemisphere holds one,672 billion tons (Petagrams) of organic carbon. If but 10 percentage of this permafrost were to thaw, it could release enough actress carbon dioxide to the atmosphere to raise temperatures an boosted 0.7 degrees Celsius (i.3 degrees Fahrenheit) by 2100.
Studying the Carbon Cycle
Many of the questions scientists still need to respond near the carbon cycle circumduct around how it is changing. The temper now contains more carbon than at any time in at least two meg years. Each reservoir of the bicycle volition change as this carbon makes its way through the wheel.
What will those changes await like? What will happen to plants as temperatures increment and climate changes? Will they remove more carbon from the atmosphere than they put back? Will they get less productive? How much actress carbon will melting permafrost put into the temper, and how much will that amplify warming? Volition ocean circulation or warming change the rate at which the sea takes up carbon? Will ocean life become less productive? How much will the bounding main acidify, and what effects will that have?
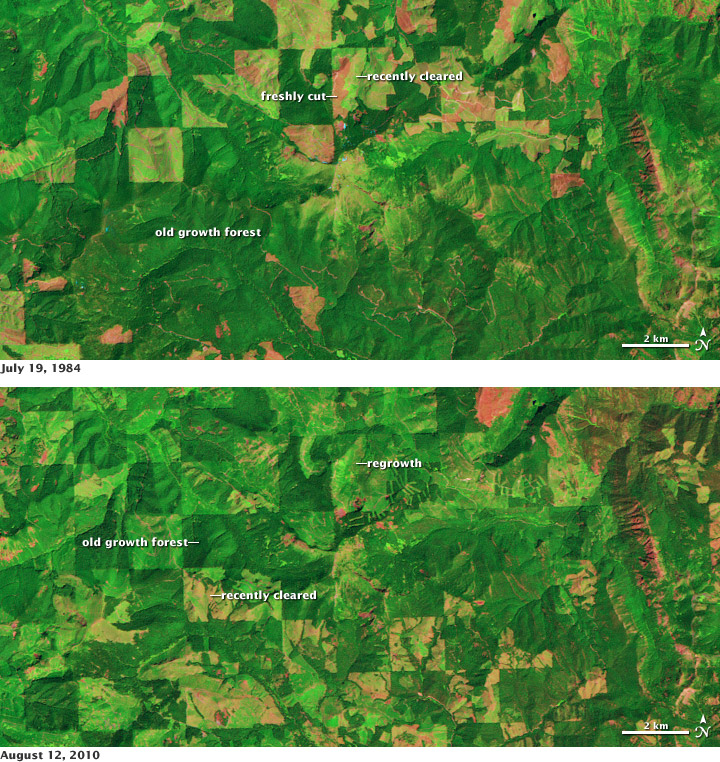
Time serial of satellite data, like the imagery available from the Landsat satellites, permit scientists to monitor changes in woods encompass. Deforestation can release carbon dioxide into the atmosphere, while forest regrowth removes CO2. This pair of false-color images shows clear cutting and forest regrowth between 1984 and 2010 in Washington State, northeast of Mount Rainier. Dark green corresponds to mature forests, red indicates blank ground or dead plant cloth (freshly cutting areas), and lite light-green indicates relatively new growth. (NASA prototype by Robert Simmon, using Landsat data from the USGS Global Visualization Viewer.)
NASA'due south role in answering these questions is to provide global satellite observations and related field observations. Every bit of early 2011, 2 types of satellite instruments were collecting information relevant to the carbon wheel.
The Moderate Resolution Imaging Spectroradiometer (MODIS) instruments, flight on NASA's Terra and Aqua satellites, mensurate the amount of carbon plants and phytoplankton turn into matter equally they grow, a measurement chosen net main productivity. The MODIS sensors also measure how many fires occur and where they burn.
Two Landsat satellites provide a detailed view of sea reefs, what is growing on state, and how land cover is changing. It is possible to see the growth of a urban center or a transformation from forest to farm. This information is crucial because land employ accounts for i-third of all man carbon emissions.
Futurity NASA satellites will continue these observations, and besides measure carbon dioxide and methane in the atmosphere and vegetation elevation and structure.
All of these measurements will help u.s. see how the global carbon cycle is changing through time. They will help us gauge the impact we are having on the carbon cycle by releasing carbon into the atmosphere or finding ways to shop information technology elsewhere. They volition evidence united states how our changing climate is altering the carbon cycle, and how the changing carbon cycle is altering our climate.
Virtually of united states, however, volition notice changes in the carbon bicycle in a more personal fashion. For u.s., the carbon cycle is the nutrient nosotros swallow, the electricity in our homes, the gas in our cars, and the atmospheric condition over our heads. We are a part of the carbon cycle, and then our decisions about how nosotros live ripple across the cycle. As well, changes in the carbon wheel volition touch on the fashion we live. As each of united states come to understand our role in the carbon cycle, the noesis empowers us to control our personal impact and to empathise the changes nosotros are seeing in the earth around us.
-
References
- Angert, A., Biraud, S., Bonfils, C., Henning, C.C., Buermann, W., Pinzon, J., Tucker, C.J., and Fung, I. (2005, August 2). Drier summers cancel out the CO2 uptake enhancement induced past warmer springs. Proceedings of the National Academy of Scientific discipline, 102 (31), 10823-10827.
- Archer, D. (2008, May). Carbon cycle: Checking the thermostat. Nature Geoscience, 1, 289-290.
- Behrenfeld, Chiliad.J., et al. (2006, December vii). Climate-driven trends in contemporary ocean productivity. Nature, 444, 752-755.
- Berner, R.A. (2003, November twenty). The long-term carbon wheel, fossil fuels and atmospheric composition. Nature, 426, 323-326.
- Bonan, Thou.B. (2008, June 13). Forests and climate modify: Forcings, feedbacks, and the climate benefit of forests. Science, 320, 1444-1449.
- Campbell, Northward.A. and Reece, J.B. (2005). Biological science. San Francisco: Pearson Benjamin Cummings. 7th ed.
- Denecke, E.J. (2002) Earth Science: The Physical Setting Hauppauge, New York: Barron's Educational Serial, Inc.
- Denman, K.L., Brasseur, G., Chidthaisong, A., Ciais, P., Cox, P.M., Dickinson, R.E., Hauglustaine, D., Heinze, C., Holland, E., Jacob, D., Lohmann, U., Ramachandran, Southward., da Silva Dias, P.L., Wofsy, S.C. and Zhang, X. (2007) Couplings Between Changes in the Climate System and Biogeochemistry. In: Climatic change 2007: The Concrete Science Basis. Contribution of Working Group I to the Quaternary Assessment Written report of the Intergovernmental Panel on Climate Change, [Solomon, S., D. Qin, M. Manning, Z. Chen, M. Marquis, Thousand.B. Averyt, One thousand.Tignor and H.L. Miller (eds.)]. Cambridge University Press, Cambridge, Great britain and New York, NY, USA.
- Doney, South.C. (2006, March). The dangers of ocean acidification. Scientific American, 58-65.
- Emsley, J. (2001). Nature's Building Blocks. Oxford: Oxford University Printing.
- Goetz, S.J., Bunn, A.One thousand., Fiske, Chiliad.J., and Houghton, R.A. (2005, September 20). Satellite-observed photosynthetic trends across boreal Due north America associated with climate and burn down disturbance. Proceedings of the National Academy of Sciences, 102 (38), 13521-13525.
- Global Carbon Project. (2010, November 21). Carbon budget 2009. Accessed May 4, 2011.
- Grosse, G., Romanovsky, V., Jorgenson, T., Anthony, 1000.W., Brown, J., and Overduin, P.P. (2011, March 1). Vulnerability and feedbacks of permafrost to climatic change. EOS, 92 (9), 73-74.
- Hansen, J., Ruedy, R., Sato, One thousand., and Lo, K. (2010, December 14). Global surface temperature alter. Reviews of Geophysics, 48, RG4004.
- Hansen, J., Nazarenko, Fifty., Ruedy, R., Sato, M., Willis, J., Del Genio, A., Koch, D., Lacis, A., Lo, M., Menon, Southward., Novakov, T., Perlwitz, J., Russel, Chiliad., Schmidt, G.A., and Tausnev, N. (2005, June 3). Earth's energy imbalance: Confirmation and implications. Science, 308 (5727), 1431-1435.
- Hardt, M.J., and Safina, C. (2010, August). How acidification threatens oceans from the inside out. Scientific American.
- Intergovernmental Panel on Climatic change. (2007). Summary for Policymakers. In: Climatic change 2007: The Physical Science Basis. Contribution of Working Group I to the Quaternary Assessment Study of the Intergovernmental Panel on Climate Change, [Solomon, Southward., D. Qin, M. Manning, Z. Chen, K. Marquis, K.B. Averyt, M.Tignor and H.L. Miller (eds.)]. Cambridge University Printing, Cambridge, United Kingdom and New York, NY, The states.
- Jouzel, J., et al. (2007). EPICA Dome C Water ice Core 800KYr Deuterium Information and Temperature Estimates. IGBP PAGES/Earth Information Center for Paleoclimatology Data Contribution Series #2007-091. NOAA/NCDC Paleoclimatology Program, Boulder CO, USA. Accessed June 13, 2010.
- Lacis, A.A., Schmidt, M.A., Rind, D., and Ruedy, R.A. (2010, October 15). Atmospheric CO2: Principal control governing Earth'due south temperature. Science, 330 (6002), 356-359.
- Lacis, A. (2010, October). CO2: The thermostat that controls Earth's temperature. NASA Goddard Institute for Infinite Studies. Accessed Dec 17, 2010.
- Le Quéré, C., Raupach, M.P., Canadell, J.G., Marland, G., et al. (2009, Nov 17). Trends in the sources and sinks of carbon dioxide. Nature Geoscience, 2, 831-836.
- Lüthi, D., K. Le Floch, B. Bereiter, T. Blunier, J.-One thousand. Barnola, U. Siegenthaler, D. Raynaud, J. Jouzel, H. Fischer, K. Kawamura, and T.F. Stocker. (2008, May 15). High-resolution carbon dioxide concentration tape 650,000-800,000 years before nowadays. Nature, 453, 379-382.
- McKinley, G.A. (2010). Carbon and Climate. Academy of Wisconsin-Madison. Accessed May 4, 2011.
- Oren, R., Ellsworth, D.S., Johnsen, K.H., Phillips, Northward., Ewers, B.Eastward., Maier, C., Schäfer, G.5.R., McCarthy, H., Hendrey, G., McNulty, S.Chiliad., and Katul, G.G. (2001, May 24). Soil fertility limits carbon sequestration by forest ecosystems in a CO2-enriched atmosphere. Nature, 411, 469-472.
- Orr, J.C. et al. (2005, September 29). Anthropogenic body of water acidification over the twenty-first century and its bear upon on calcifying organisms. Nature, 437, 681-686.
- Rothman, Fifty.S., Gordon, I.E., Barbe, A., Benner, D.C., et al. (2009, June-July). The HITRAN 2008 molecular spectroscopic database. Journal of Quantitative Spectroscopy and Radiative Transfer, 110 (nine-x), 533-572.
- Sabine, C.L. and Feely, R.A. (2007). The oceanic sink for carbon dioxide. In Greenhouse Gas Sinks, eds D.South. Reay, C.Due north. Hewitt, Grand.A. Smith, and J. Grace. CAB International.
- Sabine, C.L., et al. (2004, July 16). The oceanic sink for anthropogenic CO2. Scientific discipline, 305 (5682), 367-371.
- Schlesinger, W.H. (1991). Biogeochemistry, An Assay of Global Modify. San Diego: Academic Press.
- Schmidt, G.A., Ruedy, R.A., Miller, R.50., and Lacis, A.A. (2010, October 16). Attribution of the present-mean solar day total greenhouse upshot. Journal of Geophysical Research, 115, D20106.
- Schmidt, G. (2010, October). Taking the mensurate of the greenhouse outcome. NASA Goddard Establish for Infinite Studies. Accessed December 17, 2010.
- Schuur, E.A.G., Bockheim, J., Canadell, J.Thou., Euskirchen. Due east, Field, C.B., Goryachkin, S.V., Hagemann, S., Kuhry, P., Lafleur, P.M., Lee, H., Mazhitova, One thousand., nelson, F.E., Rinke, A., Romanovsky, V.Due east., Shiklomanov, Due north., Tarnocai, C., Venevsky, S., Vogel, J.Thousand., and Zimov, S.A. (2008, September). Vulnerability of permafrost carbon to climate change: Implications for the global carbon wheel. BioScience, 58 (8), 701-714.
- Scientific Committee of Problems of the Environs 13. (1979). Carbon in the rock wheel. In The Global Carbon Cycle, B. Bolin, E.T. Degens, S. Kempe, and P. Ketner, eds. Accessed May 4, 2011.
- Sellers, P. J., Hall, F. K., Asrar, Chiliad., Strebel, D. East., and Murphy, R. E. (1992, November 30). An Overview of the Start International Satellite Country Surface Climatology Project (ISLSCP) Field Experiment (FIFE), Journal of Geophysical Research, 97 (D17), 18,345–18,371.
- Tarnocai, C., Canadell, J.Thou., Schuur, E.A.G., Kuhry, P., Mazhitova, G., and Zimov, S. (2009, June 27). Soil organic carbon pools in the northern circumpolar permafrost region. Global Biogeochemical Cycles, 23, GB2023.
- United States Department of Energy. (2008, October nine). How fossil fuels were formed. Accessed December 17, 2010.
- United States Geological Survey. (2010, Baronial five). Volcanic gases and climate change overview. Accessed May 4, 2011.
- van der Werf, G.R., Morton, D.C., DeFries, R.Southward., Olivier, J.G.J., Kasibhatla, P.S., Jackson, R.B., Collatz, G.J., and Randerson, J.T. (2009, November). CO2 emissions from forest loss. Nature Geoscience, two, 737-738.
- Zachos, J., M. Pagani, L. Sloan, Due east. Thomas, and K. Billups. (2001, April 27). Trends, Rhythms, and Aberrations in Global Climate 65 Ma to Nowadays. Science, 292, (5517), 686-693.
- Zeebe, R.East., and Caldeira, Yard. (2008, May). Close mass remainder of long-term carbon fluxes from ice-core CO2 and bounding main chemistry records. Nature Geoscience, 1, 312-315.
brooksdeersomided.blogspot.com
Source: https://earthobservatory.nasa.gov/features/CarbonCycle
0 Response to "What Would Most Likely Happen to the Carbon Cycle if There Were No More Plants on Earth?"
Post a Comment